When are CDISC Datasets Useful for ISS?
One important objective in converting to CDISC standards is to then gain the ability to perform an ISS (integrated safety summary) analysis between multiple studies. Once the data is created in a standard format, it makes it easier to merge the data from a pool of studies since they are in the same CDISC structure. Even though the variables names and labels are standardize, the guideline does not strictly specify the length and other detail attributes. In a recent set of studies I was working on, we ran into discrepancies between two studies even though it was converted to CDISC. The following report illustrates this problem.
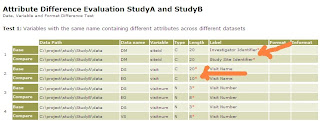
Most of the issues came about due to length differences between the two studies. This can lead to truncation if any of these key fields are merged. Other less common issues are things such as labels being different. This can be due to using different versions of the guidelines such as 3.1.1 versus 3.1.2.
I ran a %difftest macro which then revealed some difference among the attributes. This helped us standardized the data even though it was considered standard before because it was in CDISC format. Once we updated to have these attribute truly standardized, it was then useful for the ISS.
Even when things are in CDISC, it does not mean that it is useful for purposes of ISS. It is therefore recommended that consistent attributes are reviewed even on “standard” CDISC datasets before it can be useful in an integrated safety summary analysis.

Comments
Post a Comment